Radiation Medical Device Registration
Navigate the complexities of medical device regulations with confidence. We provide end-to-end support for your radiation-emitting equipment—from facility and personnel verification to seamless Regalkes submission—ensuring your products enter the market legally and efficiently.
IDAK Radiation Electromedical Amendment
We handle updating your distribution license for the radiation sub-category. This includes personnel verification (PPR & ATEM), facility documentation updates, and complete MoH liaison until your new certificate is issued.
CDAKB Radiation Scope Extension
We update your Quality Management System with radiation-specific protocols—including safety SOPs, shielding, and emergency response—to ensure your CDAKB certification covers the full Electromedical Radiation scope.
Medical Device Registration (AKL)
We manage the entire Regalkes submission process for all risk classes, covering classification, dossier preparation, administrative/technical documentation, and final approval monitoring.
Local Authorized Representative (LAR)
We serve as your official MA holder or license holder with the MoH, fulfilling mandatory in-country representation requirements without establishing a local entity.
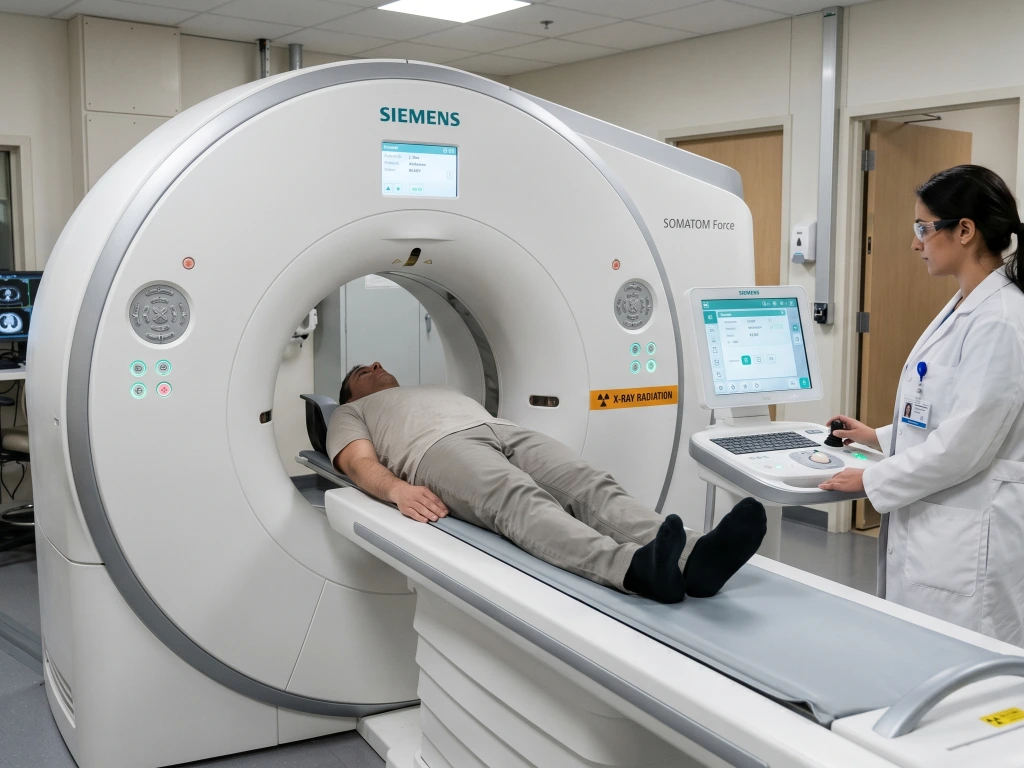
Section: Why Choose Our Radiation Medical Device Registration Service in Indonesia?
Radiation electromedical devices fall under a distinct sub-classification in Indonesia’s medical device framework. Because these devices use electrical power to emit ionizing radiation for diagnostic or therapeutic purposes, they carry stricter technical, personnel, and documentation requirements than other device categories.
Distributing these devices legally requires three core components: a Medical Device Distribution License (IDAK) with an active Electromedical Radiation scope amendment, a CDAKB certificate covering radiation-specific quality management, and product registration (AKL) through the Ministry of Health’s Regalkes system. A critical requirement unique to this category is the appointment of a Radiation Protection Officer (PPR) holding active BAPETEN certification. Without a qualified PPR on file, the IDAK radiation electromedical amendment cannot proceed.
Product Registration Indonesia coordinates all three compliance requirements under one regulatory strategy. We help international manufacturers avoid the most common causes of delay: incomplete PPR credentials, gaps in the radiation-specific QMS, and IDAK applications with incorrect sub-category scope.
当社がサポートする主要なコンプライアンス分野は次のとおりです。
- IDAK amendment to include the Electromedical Radiation sub-category
- CDAKB scope extension with radiation-specific QMS and SOPs
- Product registration dossier preparation and MoH submission for radiation device classes
シームレスなコンプライアンスサポート
エンドツーエンドのサービス
当社は、ライセンスから製品登録までの規制の全過程を管理します。.
規制に関する専門知識
当社のチームは、深い知識と経験を組み合わせて、複雑な規制環境に対応します。.
スピードと効率
当社の実績ある戦略により、インドネシア市場における登録および承認のプロセスが加速されます。.
カスタマイズされたサポート
スムーズで安心できる旅を実現するために、個別のガイダンスと専用のサポートを提供します。.
Product Registration Indonesia is Ready to Assist Your Market Entry.
Distributing radiation-emitting medical devices in Indonesia requires compliance beyond the standard medical device pathway. Our radiation medical device registration service in Indonesia supports international manufacturers with the complete Kemenkes regulatory framework, from IDAK radiation amendment and CDAKB scope extension to product registration (AKL), ensuring your X-ray equipment, CT scan systems, mammography units, fluoroscopy machines, and radiotherapy devices are legally approved and market-ready.

市場参入支援
Flexible market entry options supporting radiation medical device registration for international manufacturers entering Indonesia.
Initial Consultation
Free for a 30-minute session with our regulatory experts. We’ll review your product scope and clarify the registration pathway.
販売代理店の選択
当社は、規制および商業上の要件に準拠した適格な現地販売業者の特定と評価をお手伝いします。.
Export/Import Assistance
We manage customs clearance, import licensing, and regulatory documentation for entering the Indonesian market.
会社設立サポート
現地に拠点を設立する準備ができたら、インドネシアでの法令遵守を遵守した会社設立をお手伝いします。.
Initial Consultation
Free for a 30-minute session with our regulatory experts. We’ll review your product scope and clarify the registration pathway.
販売代理店の選択
当社は、規制および商業上の要件に準拠した適格な現地販売業者の特定と評価をお手伝いします。.
Export/Import Assistance
We manage customs clearance, import licensing, and regulatory documentation for entering the Indonesian market.
会社設立サポート
現地に拠点を設立する準備ができたら、インドネシアでの法令遵守を遵守した会社設立をお手伝いします。.
お問い合わせフォーム
弊社のチームは、お客様のビジネスニーズについてご相談に応じ、ご質問にもお答えいたします。お問い合わせフォームにご記入いただければ、1営業日以内にご返信いたします。.
弊社に連絡するその他の方法。.
よくある質問(FAQ)
What types of devices fall under the Electromedical Radiation category in Indonesia?
What Does X-ray Equipment Registration Require in Indonesia?
What is the Difference Between a Standard IDAK and a Radiation IDAK?
What is a PPR and Why is it Required for IDAK Radiation Electromedical Applications?
Is a Separate CT Scan Import License Required in Indonesia?
Can Foreign Manufacturers Register Radiation Devices Without a Local Company?
What Documents Are Typically Required for Radiation Device Registration?
Does Product Registration Indonesia Handle BAPETEN Licensing?
How Long Does the IDAK Radiation Amendment Process Typically Take?
What Happens After a Radiation Medical Device is Registered in Indonesia?

戦略的リーダーシップ
フセイン・H・マシュア博士(医学博士)
最高執行責任者(COO)であるフセイン・H・マシュア博士(医学博士)は、インドネシアの最も複雑な規制分野における製品登録およびライセンス取得において、10年以上にわたる献身的な実務経験を有しています。彼の戦略的な監督の下、当社の業務手順と顧客サービスは、実証済みの効率的なプロセスに基づいて構築されています。コンプライアンスとオペレーションの両方に精通した専門家リーダーならではの自信と明確さを持って、お客様の旅を始めましょう。.
インドネシアの医療機器メーカー向けの新しいハラール認証規則について、簡潔かつ実践的な概要を解説します。2026年までにコンプライアンスを維持し、円滑な市場参入を実現するための、主要な期限、関係当局、そして段階的な手順をご紹介します。.




